Agilent Introduces Security Pack for Analytical Laboratories
<%=company1%> Inc. (Palo Alto, CA) has introduced the Agilent Security Pack for UV-visible spectroscopy systems, which helps analytical laboratories comply with the U.S. Food and Drug Administration’s ruling on electronic data records and signatures (FDA 21 CFR Part 11).
As an add-on module for Agilent ChemStation’s advanced and dissolution testing software, the Security Pack can be installed easily on an existing ChemStation and provides users with a plug-and-go solution for access control, data security, data integrity, and audit-trails that fulfill the FDA requirement. It modifies the ChemStation’s advanced or dissolution testing software to:
- provide access control, including user setup and password administration;
- prevent loss of raw and meta data as well as unauthorized modification;
- store deleted spectra with results for later re-storage or review during an audit;
- add versioning to the ChemStation by storing all re-processed versions (each new result or method is stored in a new file); and
- follow the detailed requirements for electronic records and passwords as given by FDA 21 CFR Part 11.
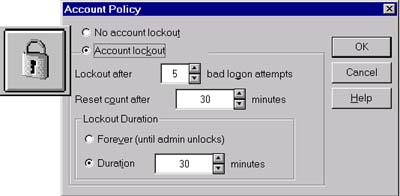
The Agilent ChemStation Security Pack helps analytical laboratories comply with FDA ruling on electronic data records and signatures.
Additional features include automated update of the run logbooks during reanalysis together with result, raw, and meta data from the analysis. Electronic signatures are added to the signature logbook, which is part of the result file. All logbook entries are generated user-independently and can be copied or printed.
The Agilent Security Pack for UV-visible spectroscopy is available worldwide and can be ordered through any Agilent Technologies sales office. The cost is $800 per software pack and delivery is estimated at two weeks from receipt of order.
For more information: Elaine Baccino, Agilent Technologies Inc., 71 Southgate Boulevard, New Castle, DE 19720. Tel: 800-227-9770. Tel: 302-633-8558.
